本サイトの内容は、医療および研究の専門知識を有する方々を対象としています。一般の方々のご利用はご遠慮ください。
SyncroPatch 384 システム
最先端の自動分注機Biomek i5にパッチクランプモジュールを統合した、画期的な自動パッチクランプシステムです。384細胞同時測定を行い、1日あたり20,000データポイントのスループットを実現します。その使いやすさとオープンなデザインで、完全な自動化とHTSへの統合をサポートします。
また32ウェルモードを使用することにより、ハイスループットスクリーニングに限らず、小規模なスクリーニングプロジェクトや学術研究など、どのようなスループットのニーズにも対応することができ、NPC-384チップのコストメリットを最大限に活用することができます。
実験に必要なウェル数を32の倍数で選択し、残りのウェルを数日間にわたって使用することができます。またプログラムを完全に自動化することにより、8時間以上の実験を無人で行うこともできます。
卓越したデータ品質と柔軟性により、SyncroPatch 384は、グローバルな製薬会社、CRO、学術機関などで採用されています。
主な特徴
- 384細胞同時測定
- 32ウェルモードにより小規模な化合物スクリーニングや研究プロジェクトにも最適化
- 高いギガシール成功率
- 高速外部溶液交換(最大110 µl/s)が可能
- 記録中の内部溶液の灌流が可能(例:細胞内カルシウム依存性のK+チャネルの記録が可能)
- 測定部と12個のデッキを個別に温度コントロール可能(温度範囲:10~37℃)
- カレントクランプによる活動電位の測定も可能
- シングル & マルチホールチップを使用可能(自社製造)
- プログラムの自動化により、約8時間の無人運転が可能
- 濃度解析のために測定後の溶液を回収することが可能
- 優れたサービスとサポートを提供します

ハードウェアー/ソフトウェア
- ナニオンのパッチクランプチップは、マニュアルパッチクランプで使用されるガラスキャピラリーと同様にホウケイ酸ガラス製のため、低キャパシタンス、低化合物吸着、優れたパッチ/シール特性を実現しています
- オンデッキでの化合物プレートの準備 - 化合物の準備のために別の自動分注機を使用したり、手作業で化合物プレートを準備する必要はありません。
- 流路タイプのチップと比較して、溶液と壁面の接地面積が低いため、化合物の吸着が低い
- 化合物暴露 & wash回数の制限なし
- グリッパーとピペッティングヘッドが独立して稼働するため、例えば化合物の蓋を外す際にピペッティングチップを外す必要がありません
- 少量の化合物で実験が可能
- 強力な解析ソフトウェア(フレキシブルなバッチ解析、IC50計算、IV解析、Genedata Screenerとの互換性あり)
- z-primeとヒートマップにより、データの概要を迅速かつ視覚的に把握することが可能
- 設定、編集、分析が簡単にできます
- スタンダードとアドバンスの異なる操作モードにより、実験パラメータの柔軟性を維持しながら、シンプルに操作できます
- スループットの選択 - 32ウェルモードでは、実験に適したスループットを常に選択できます
アプリケーション
- 電位依存性 & リガンド依存性チャネルに対応
- 少ない細胞量で実験が可能 – 幹細胞や初代細胞にも適用可能
- 様々なイオンチャネルに適用可能
- CiPA validation studyにおいて様々なデータを提供
- 創薬のあらゆるフェーズに対応
- データを様々なフォーマットでエクスポートし、外部ツールで簡単に解析可能
- 様々なCROアッセイに対応できる柔軟性
特徴と仕様
Technical Specifications of the SyncroPatch 384

SyncroPatch 384: Standard Delivery Package
The SyncroPatch 384 includes:
- Biomek i5 with a 384-pipettor arm, gripper and temperature-controlled deck positions
- Temperature-controlled patch clamp module
- Amplifier (384 channels) incl. current clamp
- Windows 10 OS with PatchControl 384 and DataControl 384 software suite
- Guided Labware Setup and Method Launcher
- Temperature-controlled cell hotel
- Barcode scanner
- NPC-384 borosilicate recording chips
- Optional service plans for unmatched
ソフトウェア
PatchControl 384

DataControl 384: The Analysis Software
専用チップ
NPC-384
The NPC-384 chip is the cost-efficient and high quality consumable for the SyncroPatch 384. It is produced and quality-assured in-house at Nanion headquarters in Munich. Different types of NPC-384 chips are available which should be chosen depending on cell size and application.

Material
The borosilicate glass slide with the patch aperture is encased in a 384 well plate forming wells where the cells and external solutions are delivered. The design of the chip allows perfusion of the internal solution during an experiment.
Features
Each NPC-384 chip contains 384 recording chambers. These sites can be used all at one time or in 32-well mode, parts of the chip can be used in multiples of 32, and the remaining parts used over several days, without a reduction success rate. One chip can be measured on the SyncroPatch 384 and 25 chips can be stacked in the robot for unattended experiments. The open design of the chip makes sample collection and subsequent verification of compound concentration. Additionally, the number of exchanges of either the internal or the external solution is unlimited. NPC-384 chips can be purchased as single hole per well for GOhm seals, or multi-hole per well to increase measured current amplitude and improve success rate.
Available chip types
- "NPC-384, 1x medium resistance": One hole per well (Order # 221102)
- "NPC-384, 1x medium resistance plus": One hole per well (Order # 221104)
- "NPC-384, 4x medium resistance": 4 holes per well (Order # 221402)
- "NPC-384, 1x high resistance": One hole per well (Order # 221101)
- "NPC-384, 4x high resistance": 4 holes per well (Order # 221401)
- "NPC-384, 1x low resistance": One hole per well (Order # 221103)
- "NPC-384, 4x low resistance": 4 holes per well (Order # 221403)
- "NPC-384, 8x": 8 holes per well (Order # 221801)
試薬
Buffers and Solutions for the SyncroPatch 384/384i/768i
Reliable buffer solutions are critical for any electrophysiological application. Our goal, therefore, is to provide high-class recording solutions that leave you in no doubt about quality and stability. Our quality assurance includes chemical tests as well as tests on our patch clamp systems of each lot. Our buffers are shipped with the corresponding "Certificates of Analysis" and "Material Safety Data Sheets" (MSDS).
Available buffers and solutions
- "External Standard", 500 mL: (Order # 08 3001)
- "External Standard Ca 10", 500 mL: (Order # 08 3012)
- "External NMDG 60", 500 mL: (Order # 08 3004)
- "External NMDG 60 Ca 10", 500 mL: (Order # 08 3011)
- "External [-] Ca2+ [-] Mg2+", 500 mL: (Order # 08 3003)
- "Internal CsF 110", 500 mL: (Order # 08 3008)
- "Internal KF 110", 500 mL: (Order # 08 3007)
- "Washing solution", 5 L: (Order # 08 3010)
データ&アプリケーション
32- well mode for smaller screens or academic investigations
SyncroPatch 384 data and applications:
Cells were kindly provided by SB Drug Discovery
An exemplary 32-well Mode Experiment. A small fraction of the chip can be used at a time, which is ideal for smaller compound screens.
Consecutive experiments of 32-wells on the same NPC-384 patch clamp chip over multiple days. Success rate and accurate pharmacology remains stable over 8 days as shown in the figure. Nav1.5 recordings in the presence of increasing Mexiletine concentrations.

AMPA Receptor (GluA2) - Activation by Glutamate
SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by SB Drug Discovery.
The AMPA receptor (GluA2) was activated using different concentrations of glutamate (1 µM - 100 µM). Measured on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384), the whole cell patch methodology and multi-hole chips were used.
The lower two images are displaying screenshots of single cell currents after repetitive glutamate applications:
Left: The same concentration of Glutamate was applied three times.
Right: Four different Glutamate concentrations were applied in a cumulative manner.

AMPA Receptor (GluA2) - Cumulative Concentration Response
SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by SB Drug Discovery.
The AMPA receptor (GluA2)was activated by increasing concentrations of glutamate on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384). L-glutamate was applied for approximately 500 ms in increasing concentrations (A) and a cumulative concentration response curve for glutamate was constructed for 222 wells (C).
The online analysis values peak amplitude and area under the curve (AUC) are shown versus time in Panel B. The fast activation of GluA2 could be captured at higher concentrations (inset; 1 mM).

AMPA Receptor (GluA2) - Pharmacology
SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by SB Drug Discovery.
The AMPA receptor (GluA2) was analyzed using different positive and negative allosteric modulators (CNQX, LY404187, LY395153, CP465022, Cyclothiazide). After activating the receptor by application of Glutamate, the modulating compound plus glutamate was applied afterwards. Measured on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384), the whole cell patch methodology and multi-hole chips were used.
The lower images on the left hand side are displaying a screenshot of a current after application of the positive modulator LY404187. The EC50 was determined as 379 nM.

Cardiac Ion Channels - Pharmacology of Sotalol
CardioExcyte 96 and SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by Charles River and Cellular Dynamics.
The image on the left hand side displays the results of the blocking effect of Sotalol on hERG. The result is in good agreement with manual patch clamp data (Crumb et al., 2016). The compound induced arrhythmia when iPSC-CM were exposed to a minimum concentration of 10 µM. Arrhytmic events were both detected in field potential recordings as well as in the impedance based contractility measurements.

CaV1.2 - Current Voltage Relationship
SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells kindly provided by Charles River.
CaV1.2 expressed in CHO cells recorded on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384). A The screenshot shows the data acquisition and analysis software used on the SyncroPatch 384PE. The online analysis values are shown for a current-voltage experiment. B The raw traces from an example cell elicited by depolarizing steps from -60 mV to 40 mV in 10 mV increments from a holding potential of -80 mV are shown. C The normalized current-voltage plot for an average of 272 cells. A Boltzmann equation fit revealed a V0.5 of activation of -4.8 mV.

CaV1.2 - Pharmacology of Nifedipine, using the CiPA protocol
SyncroPatch 384PE (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by Charles River.
Screenshots of the PatchControl 384 software showing hCaV1.2/β2/α2δ1 current traces in response to the CiPA voltage step protocol and the corresponing current-voltage relationship plot. Measured on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384) using perforated patch methodology (Escin) and multi-hole chips (4 holes per well), the success rate of valuable data for the analysis was 94%. The IC50 value of Nifedipine was determined as 106 nM.

CaV1.2 - Stable recording from frozen stock cells
Cells were kindly provided by Charles River.

ClC-1 - Current-voltage plot
SyncroPatch 384i (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by Charles River.
Activation of hClC-1 tail currents expressed in CHO cells recorded on the SyncroPatch 384i (a predecessor model of SyncroPatch 384). A pre-pulse voltage step to +60 mV was followed by voltage steps from -120 mV to +80 mV for 300 ms (increasing in 20 mV steps) and the tail current was measured at the subsequent step to -100 mV. Out of a possible 384 wells, all 384 wells were used for the IV analysis

ClC-1 – Inhibition by 9-AC
SyncroPatch 384i (a predecessor model of SyncroPatch 384) data and applications:
Cells were kindly provided by Charles River.
Tail currents of ClC-1 expressed in CHO cells were inhibited by increasing concentration of 9-AC. A single concentration of 9-AC was added to each well and the concentration response curve constructed over multiple wells. The IC50 was calculated to be 6.3 µM for an average of 352 wells. The average current traces are also shown.

GABAA Receptor (α1β2γ2) - Success Rates
Cells were kindly provided by Bsys.

GluA2 activation at 110µl/s – speed is key
SyncroPatch 384 data and applications:
Cells were kindly provided by SB Drug Discovery
The AMPA receptor (GluA2) was activated using increasing concentrations of glutamate. Measured on the SyncroPatch 384 the whole cell patch methodology and multi-hole chips were used. The faster you apply the ligand, the shorter is the Time to Peak, this means pipetting speed is relevant for accurate pharmacology. The IC50 of Glutamate at 110 µl/s was 460 µM.

Glycine Receptor (GlyRa1) - Reproducible Current Recordings

hERG - Pharmacology at Physiological Temperature using the CiPA Protocol
Cells were kindly provided by Charles River.

hERG - Pharmacology using the CiPA Protocol
Cells were kindly provided by Charles River.

hERG - recordings with great stability using the CiPA step ramp protocol
Cells were kindly provided by Charles River.

hERG - Stable Recordings with Accurate Pharmacology
Cells were kindly provided by Charles River Laboratories.

hERG and Temperature Control
Cells were kindly provided by Charles River Chantest

KCa1.1 (BK) - High throughput study
Data kindly provided by Sharan R. Srinivasan1 and Vikram G. Shakkottai1,2
1Department of Neurology, University of Michigan, Ann Arbor, MI 48109;
2Molecular and Integrative Physiology, University of Michigan, Ann Arbor, MI 48109.

KCa3.1 (SK4) - Activation by Perfusion of free internal Calcium
Cells were kindly provided by Charles River.

Kir2.1 - Pharmacology of Barium
Cells were kindly provided by Charles River.

KV1.3 - Pharmacology with High Success Rate
Cells were kindly provided by Evotec.

KV1.5 - Dose response curve of 4-AP
Cells were kindly provided by Charles River.
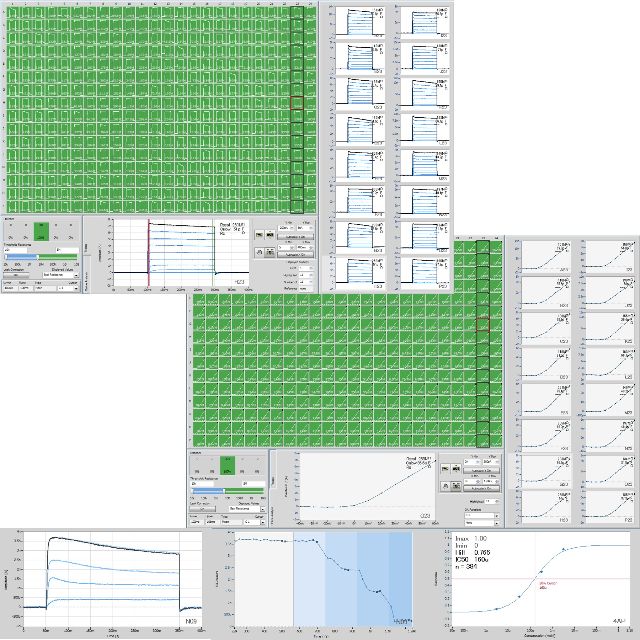
KV4.3 - Pharmacology of Metropolol Tartrate, using the CiPA Protocol
Cells were kindly provided by Charles River.
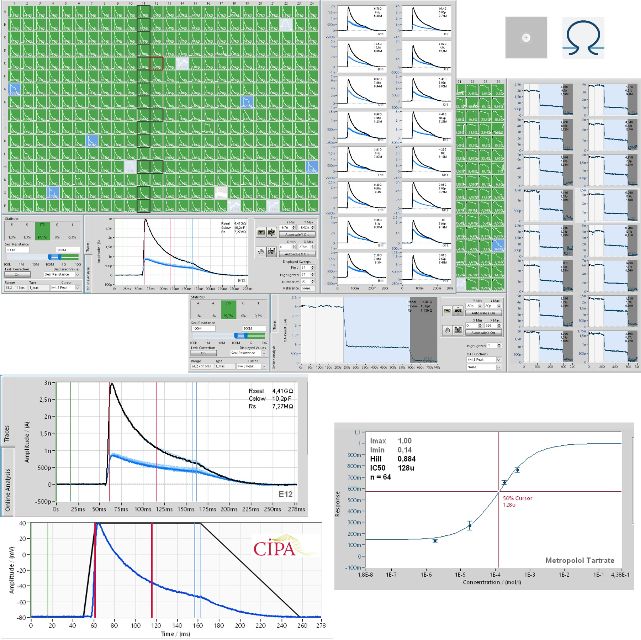
KV4.3 - Pharmacology of Quinidine
Cells were kindly provided by Charles River.

KV4.3/KChIP2 - Dose-response curve of Flecanaide
Cells were kindly provided by Charles River.

KV7.1 (KVLQT) - Dose-response curve
Cells were kindly provided by Charles River.
Screenshots of the PatchControl 384 software showing KV7.1/KCNE (KVLQT/minK) current traces in response to a voltage step protocol and the corresponing current-voltage relationship plot. Using the perforated patch methodology (Escin) in combination with multi-hole chips (4 holes per well), stably transfected cells were measured on the SyncroPatch 384PE (a predecessor model of SyncroPatch 384). The IC50 value of Chromanol 293B was determined as 3.82 µM. The success rate of completed experiments was 100%.

NaV1.5 - Current Voltage Relationship
Cells were kindly provided by EMD Millipore.

NaV1.5 - Inactivation Protocol
Cells were kindly provided by EMD Millipore.

NaV1.5 - Late Current Analysis using the CiPA Protocol
Cells were kindly provided by Charles River.

NaV1.5 - Lidocaine Dose Response
Cells were kindly provided by EMD Millipore

NaV1.7 - Accurate Voltage Clamp
Cells were kindly provided by Anaxon AG.

NaV1.7 - Frequency Dependent Inhibition
Cells were kindly provided by Anaxon.

NaV1.7 - Pharmacology of Lidocaine
Cells were kindly provided by Anaxon.

NaV1.8 - I/V Characteristics

NaV1.8 - State Dependent Inhibition
Cells were kindly provided by Charles River.

NaV1.9 - Pharmacology
Cells were kindly provided by Icagen.

NMDA NR1/NR2A - Activation and Modulation
Cells were kindly provided by B'Sys.

P2X2/ P2X3 - Activation by ATP on the SyncroPatch 384PE
Cells were engineered and kindly supplied by Axxam

P2X2/ P2X3 - Block by suramin on the SyncroPatch 384PE
Cells were engineered and kindly supplied by Axxam

P2X2/ P2X3 - High throughput screening on the SyncroPatch 384PE
Cells were engineered and kindly supplied by Axxam

Piezo1 in Neuro2A cells - activation by Yoda1
Piezo1 channels endogenously expressed in Neuro2A cells were investigated on the SyncroPatch 384PE (a predecessor model of the SyncroPatch 384). A Screenshot of the PatchControl 384 software during an experiment. B Statistical analysis of the currents at -100mV (left) and at 80 mV (right). 140 out of 384 Neuro2A cells (37%) passed the quality criteria and 85 cells (60% of the valid cells) were considered as Yoda1 responders.
Data from Rotordam et al, 2019.

Piezo1 in red blood cells - activation by Yoda1
Current response of Piezo1 activated by Yoda1 in patient cells with the novel PIEZO1 mutation (R2110W) compared to healthy red blood cells (RBCs). Shown are raw data traces (top) and statistical analysis of all measured cells, independent of their response to Yoda1 (bottom).
Data from Rotordam et al, 2019.

Piezo1 in red blood cells - Hereditary Xerocytosis
Whole-cell recordings of ion currents from RBCs of healthy donors and Hereditary Xerocytosis patients. Different mutations in the PIEZO1 gene were compared with controls. Aa The P50.2 mutation resulted in current conductance that was unchanged compared with transport controls, but showed increased conductance compared with general controls (Ab). The mutation P52.1 showed decreased conductance compared with transportation controls (Ba) and general controls (Bb).
Data from Petkova-Kirova et al, 2019.

TRPA1 - Activation by carvacrol
Cells were kindly provided by AcCELLerate.

TRPA1 - Inhibition by A967079 and AMG0902
Cells were kindly provided by AcCELLerate.

TRPM8 and Temperature Control
Cells were kindly provided by Charles River Chantest

インタビュー&ケーススタディー
Dr. David Dalrymple - Statement about the SyncroPatch 384PE
Business Development Director at SB Drug Discovery
Prof. Al George - A New Era is Emerging for Ion Channel and Channelopathy Research
“... the SyncroPatch has revolutionized our ability to determine the functional consequences of hundreds of human ion channel variants, which could be considered one of the most significant recent advances in channelopathy research.”
Prof. Dr. Al George
Professor and Chair of Pharmacology at Northwestern University Feinberg School of Medicine, Chicago, IL, USA
Download the full customer case study here:
SyncroPatch 384i (a successor of SyncroPatch 384PE) Customer Case Study: (3.1 MB)
Prof. Dr. Al George, Professor and Chair of Pharmacology at Northwestern University Feinberg School of Medicine, Chicago, IL, USA
Prof. Al George - Statement about the SyncroPatch 384PE
“We are extraordinarily excited about installing the first SyncroPatch 384PE (a predecessor model of SyncroPatch 384i) in an academic setting in North America. The enormous throughput, intuitive software and robust liquid handling capabilities along with superior seal quality, stability and high success rates convinced us to purchase the instrument. The SyncroPatch 384PE will enable us to perform detailed high throughput analysis of genetic variants in human ion channels at a previously unobtainable scale, and will form the cornerstone of a new HTS facility we are building. We also look forward to upgrading to 768 wells in the near future.“
Dr. Al George
Professor and Chair of Pharmacology at Northwestern University Feinberg School of Medicine, Chicago, IL, USA
Prof. Jamie Vandenberg - Statement about SyncroPatch Technology
Prof. Dr. Jamie Vandenberg, co-deputy director and head of cardiac electrophysiology
The Victor Chang Cardiac Research Institute, Australia
Nina Braun - Statement about the SyncroPatch 384i
“I measured CRCs for activation and steady-state desensitization, as well as peptide modulation of the channel, and got fantastic support from Søren Friis both with technical questions and assay design .The SyncroPatch and its software are easy to use and allow for versatile assay design. The team at Nanion goes out of their way to help with all questions and requests that come up, and they host fantastic user meetings for idea exchange. After four years using the SyncroPatch, I can highly recommend it.”“
Nina Braun
University of Copenhagen
チュートリアル
Basic principles of external solution exchange and compound addition
Also applies to the SyncroPatch 384
ウェビナー&動画
14.10.2020 | Webinar: Development and validation of ASIC1a ligand-gated ion channel drug discovery assays on automated patch clamp platforms
15.10.2020 | Webinar: Turning Cells into Reagents
15.10.2020 | Webinar: ICH S7B best practices considerations - New Q&As and Benchmarking best practices
15.10.2020 | Webinar: Benchmarking best practices and calibration standards for HTS hERG recordings for improved proarrhythmic assessment
Speakers:Dr. Alison Obergrussberger (Nanion Technologies)
This is an on-demand webinar from Nan]i[on and Friends 2020.
16.10.2020 | Webinar: Emerging Role of LRRC8 Volume-Regulated Anion Channels in the Skin
Dr. Oliver Rauh (Technical University of Darmstadt)
Giustina Rotordam (Nanion Technologies)
2020 - What is the Syncropatch 384PE and what are the benefits of the Syncropatch 384PE?
2019 - SyncroPatch 384i Product Video
The SyncroPatch 384i – is a giga-ohm seal HTS automated patch clamp platform based on the newly introduced and state-of-the art Biomek i5 liquid handler.
It provides effortless ion channel screening coupled with unmatched flexibility, ease-of-use and reliability. The SyncroPatch 384i builds on the success of the SyncroPatch 384PE, which has been globally established as the preferred automated patch clamp workhorse in Pharma, Biotech, CRO and academia.
2018 - HTS Phase I study: an update on progress of the CiPA Ion Channel Work Stream using the SyncroPatch 384PE and Patchliner
Tim Strassmaier, Nanion Technologies Inc. USA
Source:
Webinar: "CiPA study: Bridging ion channel and myocyte data", September 12, 2018
2018 - Biophysical and Pharmacological Characterization of Voltage-Gated Sodium Channels Involved in Pain Pathways
Dr. Markus Rapedius, Senior Scientist, Nanion Technologies